- HOME
- VENUE
- RSVP
- REGISTRY
- CONTACT
- Zombieland movie similiar
- Rtmp server ubuntu 17-10
- Skyrim mods reddit
- Urgrove movie site
- Driver usb tv stick
- Gta san andreas mod packs
- Download free turbo c for windows 7 32 bit
- Dragon ball z tenkaichi tag team ppsspp multiplayer
- Cvs delphi
- Half life problems
- Ercom aircom
- Ansi asq z1 4 2008 pdf
- I will give camtasia studio 8 lifetime version
- Mario kart 8 iso pc
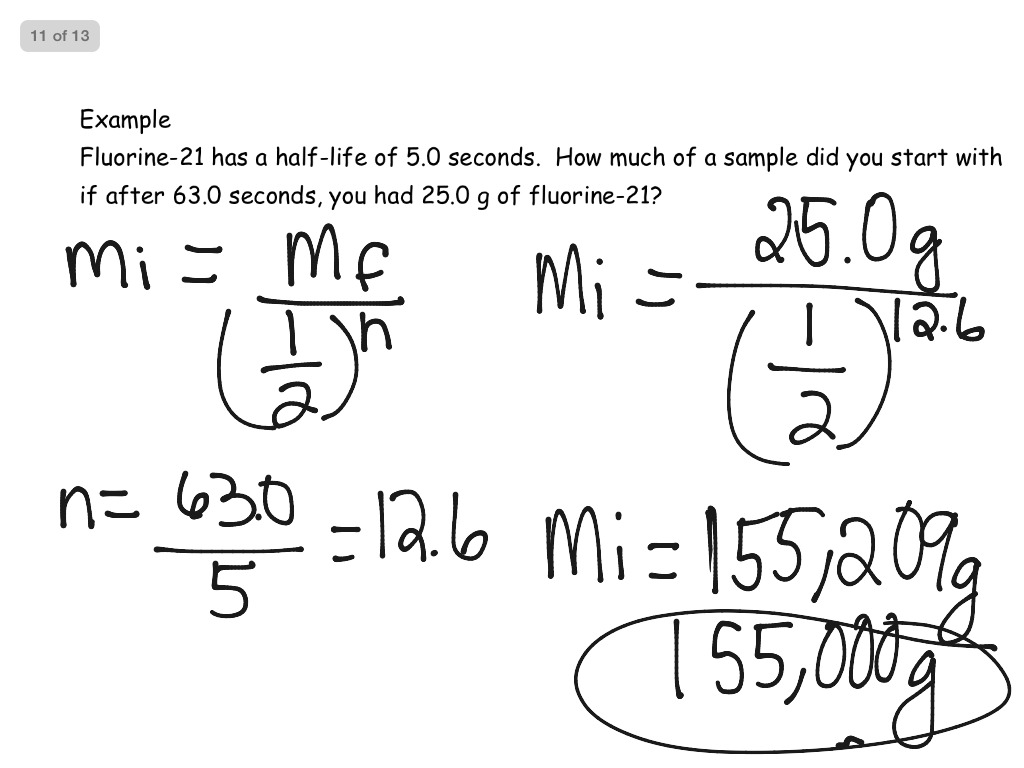
The term "half-life" is almost exclusively used for decay processes that are exponential (such as radioactive decay or the other examples above), or approximately exponential (such as biological half-life discussed below). In a first-order reaction the half-life of the reactant is ln(2)/ λ, where λ is the reaction rate constant.

Nevertheless, when there are many identical atoms decaying (right boxes), the law of large numbers suggests that it is a very good approximation to say that half of the atoms remain after one half-life. Note that after one half-life there are not exactly one-half of the atoms remaining, only approximately, because of the random variation in the process. įor example, the image on the right is a simulation of many identical atoms undergoing radioactive decay. In other words, the probability of a radioactive atom decaying within its half-life is 50%. Instead, the half-life is defined in terms of probability: "Half-life is the time required for exactly half of the entities to decay on average". For example, if there is just one radioactive atom, and its half-life is one second, there will not be "half of an atom" left after one second. In that case, it does not work to use the definition that states "half-life is the time required for exactly half of the entities to decay". Note the consequence of the law of large numbers: with more atoms, the overall decay is more regular and more predictable.Ī half-life usually describes the decay of discrete entities, such as radioactive atoms. The number at the top is how many half-lives have elapsed. Simulation of many identical atoms undergoing radioactive decay, starting with either 4 atoms per box (left) or 400 (right). The accompanying table shows the reduction of a quantity as a function of the number of half-lives elapsed. Half-life is constant over the lifetime of an exponentially decaying quantity, and it is a characteristic unit for the exponential decay equation. Rutherford applied the principle of a radioactive element's half-life to studies of age determination of rocks by measuring the decay period of radium to lead-206. The original term, half-life period, dating to Ernest Rutherford's discovery of the principle in 1907, was shortened to half-life in the early 1950s. The converse of half-life is doubling time. For example, the medical sciences refer to the biological half-life of drugs and other chemicals in the human body. The term is also used more generally to characterize any type of exponential or non-exponential decay. The term is commonly used in nuclear physics to describe how quickly unstable atoms undergo radioactive decay or how long stable atoms survive. The time in which half of the original number of nuclei decay is defined as the half-life, t 1 / 2 t 1 / 2 size 12.Half-life (symbol t 1⁄2) is the time required for a quantity to reduce to half of its initial value. Why use a term like half-life rather than lifetime? The answer can be found by examining Figure 31.21, which shows how the number of radioactive nuclei in a sample decreases with time.

In this section we explore half-life and activity, the quantitative terms for lifetime and rate of decay. This means they have shorter lifetimes, producing a greater rate of decay. For example, radium and polonium, discovered by the Curies, decay faster than uranium. However, some nuclides decay faster than others.